Bacterial infections during fuel ethanol fermentations can negatively impact the fermentation process and lower ethanol yields. Lactic Acid-Producing Bacteria (LAB), such as Lactobacillus, Lactococcus, Weissella and Pediococcus, are a common source of contamination due to their ability to grow in the harsh conditions, low pH, high temperatures, and high concentrations of ethanol, present during fermentation. LAB are problematic as they not only compete with the yeast for nutrients and fermentable sugars, they also produce lactic and acetic acids that influence yeast metabolism. Ultimately, untreated bacterial infections cost plants time and money. Antibiotics, such as the β-lactam antibiotic penicillin G and the streptogramin antibiotic virginiamycin, are commonly used during fermentations to control bacterial growth and support robust ethanol yields.
In response to a competitive antibiotic market and increasing antibiotic resistance, antibiotic combinations, particularly combinations of penicillin G and virginiamycin, have recently been employed. However, there is little evidence supporting the use of a penicillin G/virginiamycin combination treatment during fuel ethanol fermentations. When drugs such as antibiotics are applied concurrently, drug interactions must be considered. Drug interactions can be classified as additive, synergistic, or antagonistic (Figure 1). An additive interaction is when two drugs do not interact with each other and the response to the combination is the sum of the individual drug responses. Drugs are synergistic when use of the two drugs together creates a greater response than either drug individually. In direct contrast, drugs are antagonistic when the combination leads to a lesser or less successful response compared to the individual components.

Figure 1. Graphical representation of possible interactions between drug A and drug B: additive (blue), antagonistic (red), and synergistic (green).
Figure adapted from
Synergistic drug pairs are rare occurring in only 4-10% of drug pairs tested, and if the drugs target different metabolic pathways the chance of antagonism increases
To explore the relationship between penicillin and virginiamycin when used as a combination treatment during ethanol fermentation, we compared the efficacy of each individual antibiotic and various combinations of the two against both lab bacterial strains and isolates collected from operating ethanol plants. Pediococcus pentosaceus, a common bacteria found in ethanol fermentations, was grown in the presence of either no antibiotic, penicillin G or virginiamycin alone, or 1:1, 2:1, or 3:1 ratios of penicillin to virginiamycin. Isobolograms, a common graphical tool used to analyze drug combination results, were used to determine whether the drug combinations were synergistic (more effective), additive (neutral), or antagonistic (less effective)
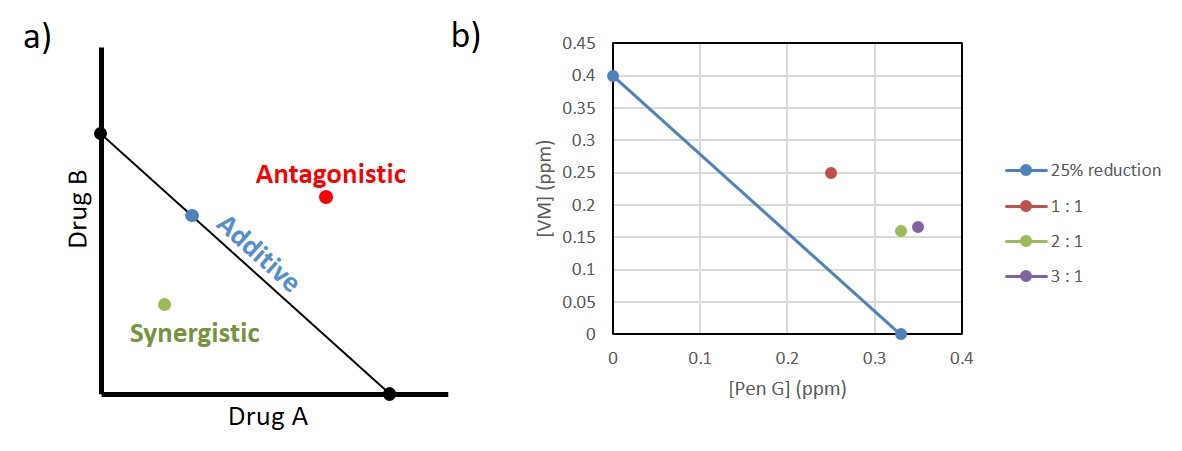
Figure 2. Investigating drug synergy and antagonism using isoboles. a) Example of an isobole where concentration of drug A and drug B give an equal response. Any point on the line is an example of additive or neutral drug interactions. Any point below the line is synergistic and any point above the line is antagonistic. b) Isobole of Pediococcus growth response to penicillin G (PenG) and virginiamycin (VM). All points on the graph represent the concentration of the specified antibiotic that achieves 25% reduction in growth. All antibiotic combinations tested (1:1 (red), 2:1 (green), and 3:1 (purple) penicillin to virginiamycin) showed antagonistic interactions.
In addition to testing a lab strain, we also tested fourteen isolates from nine different operating ethanol plants over the past couple years. As with the lab bacterial strain, isolates were grown in the presence of either no antibiotic, each individual antibiotic, or various combinations. For analysis of the results, we chose to focus on 40%, 25%, and 10% growth reduction responses as the average antibiotic concentration required fell within the typical range of antibiotic doses used in the field, 1.8, 1, and 0.5 ppm respectively (Figure 3). While there were a few examples of synergy seen with the penicillin/virginiamycin combinations, the results overall showed a significant trend toward antagonism. This becomes especially true at lower antibiotic concentrations. When looking at the results that correspond to a 0.5 ppm average antibiotic dose, twelve of the fourteen isolates reacted antagonistically with the combination products. Our results show that greater doses of the combination products would be required to achieve the same antibacterial activity of either penicillin or virginiamycin alone. Antibiotic combinations are less effective at controlling bacterial growth compared to the individual antibiotics.
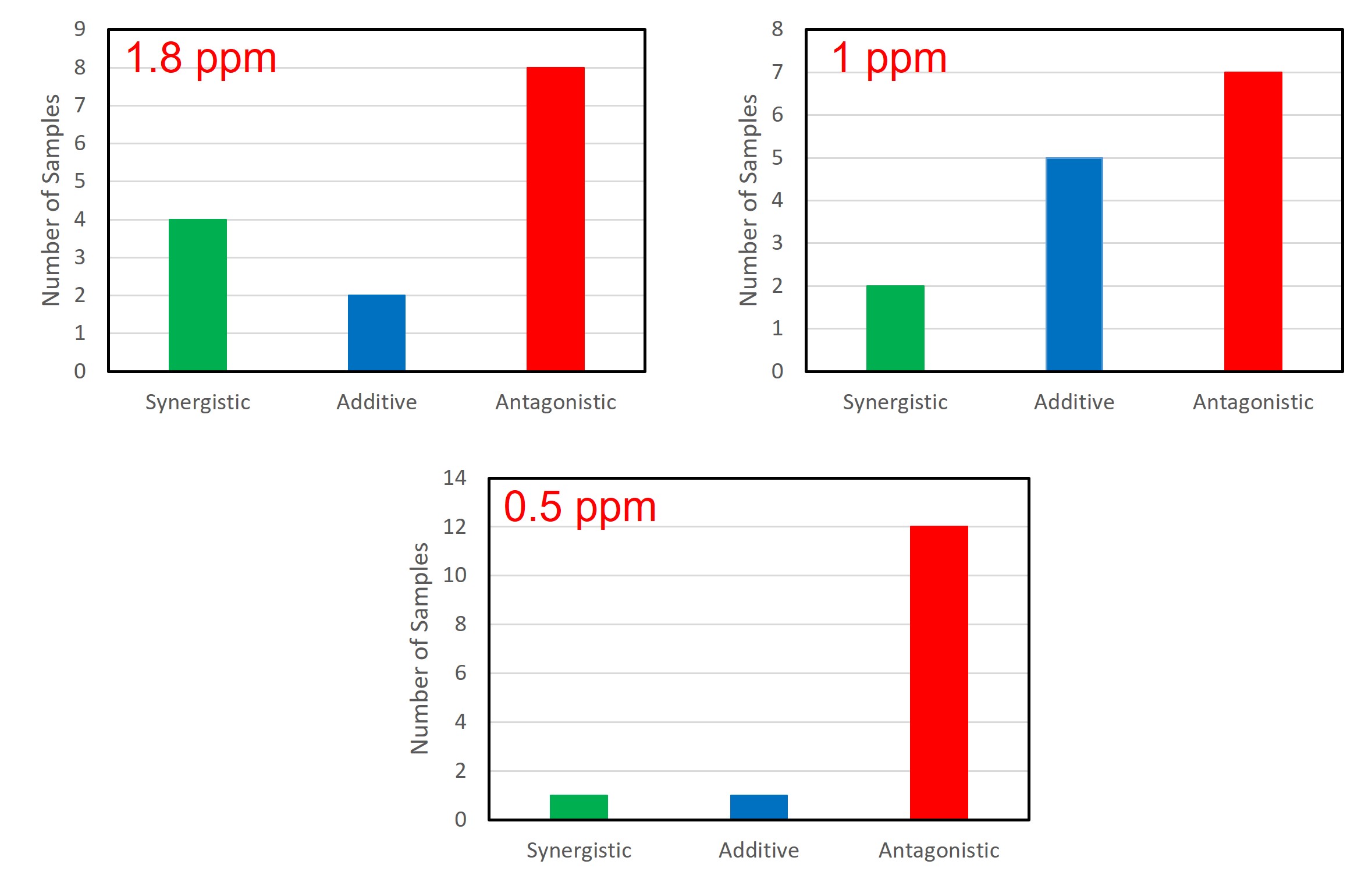
Figure 3. Responses to antibiotic combinations by isolates from operating ethanol plants. Plant isolates respond primarily antagonistically especially at lower antibiotic concentrations. The average antibiotic concentration required to achieve the specified growth reduction is shown in red.
Development of antibiotic resistance is a major concern for the continued use in fuel ethanol production. With so few antibiotics approved for use, development of multi-drug resistant bacterial strains would be extremely detrimental. Combination products are applying two of the available antibiotics at the same time increasing the risk of multi-drug resistance
To further investigate how the antibiotic application method influences development of resistance, Pediococcus pentosaceus was once again grown in the presence of either penicillin, virginiamycin, a 1:1 penicillin to virginiamycin combination, or an alternating schedule of penicillin followed by virginiamycin. Pediococcus was grown in a series of wells with a gradient of antibiotic. Each day, cells from the highest concentration of antibiotic that still saw growth were used to inoculate a fresh set of media supplemented with an increased gradient of antibiotics (Figure 4). After a single day of growth, all antibiotic treatment methods tested showed growth at ≤ 0.7 ppm antibiotic. After twelve days, both penicillin and virginiamycin exposed cells showed significant gain of resistance, showing growth at 7.5 ppm and 15 ppm respectively. The 1:1 penicillin/virginiamycin blend exposed cells developed resistance to 3.8 ppm antibiotic. Alternating penicillin and virginiamycin was the most successful at limiting resistance development; these cells developed resistance to only 1.3 ppm antibiotic. Only the alternating method kept antibiotic resistance at, or below typical antibiotic doses used in fuel ethanol fermentation.
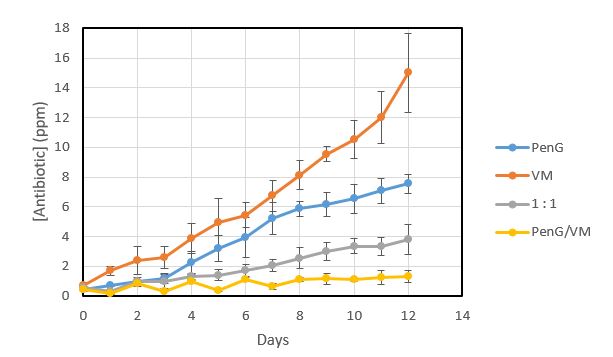
Figure 4. Comparison of the development of antibiotic resistance in Pediococcus when dosed with penicillin G (blue), virginiamycin (orange), a 1:1 combination of penicillin and virginiamycin (grey), and alternating penicillin for a day followed by virginiamycin for a day (yellow).
Overall, our results show that penicillin G and virginiamycin when applied simultaneously act overwhelmingly antagonistically against both lab bacterial strains and field isolates. Antibiotic combination products are more expensive, generally less effective, and increase the likelihood of multi-drug resistance. In addition to thorough CIP, use of a single antibiotic with periodic switching to an alternative antibiotic with a different mode of action provides the most effective bacterial control while limiting development of multi-drug resistance.
References
Bischoff, K. M., Skinner-Nemec, K. A., & Leathers, T. D. (2007). Antimicrobial susceptibility of Lactobacillus species isolated from commercial ethanol plants. Journal of Industrial Microbiology and Biotechnology, 739-744.
Bollenbach, T. (2015). Antimicrobial interactions: Mechanisms and implications for drug discovery and resistance evolution. Current Opinion in Microbiology, 1-9.
Cokol, M., Chua, H. N., Tasan, M., Mutlu, B., Weinstein, Z. B., Suzuki , Y., . . . Roth, F. P. (2011). Systematic exploration of synergistic drug pairs. Molecular Systems Biology, 544.
Johansen, H. K., Jensen, T. G., Dessau, R. B., Lundgren, B., & Frimodt-Moller, N. (2000). Antagonism between penicillin and erythromycin against Streptococcus pneumoniae in vitro and in vivo. Journal of Antimicrobial Chemotherapy, 973-980.
Kim, S., Lieberman, T. D., & Kishony, R. (2014). Alternating antibiotic treatments constrain evolutionary paths to multidrug resistance. PNAS, 14494-14499.
Pena-Miller, R., Laehnemann, D., jansen, G., Fuentes-Hernandez, A., Rosenstiel, P., Schulenburg, H., & Beardmore, R. (2013). When the most potent combination of antibiotics selects for the greatest bacterial load: The smile-frown transition. PLOS Biology, e1001540.
Tallarida, R. J. (2012). Revisiting the isobole and related quantitative methods for assessing drug synergism. Perspectives in Pharmacology, 2-8.
Yeh, P., Tschumi, A. I., & Kishony, R. (2006). Functional classification of drugs by properties of their pairwise interactions. Nature Genetics, 489-494.